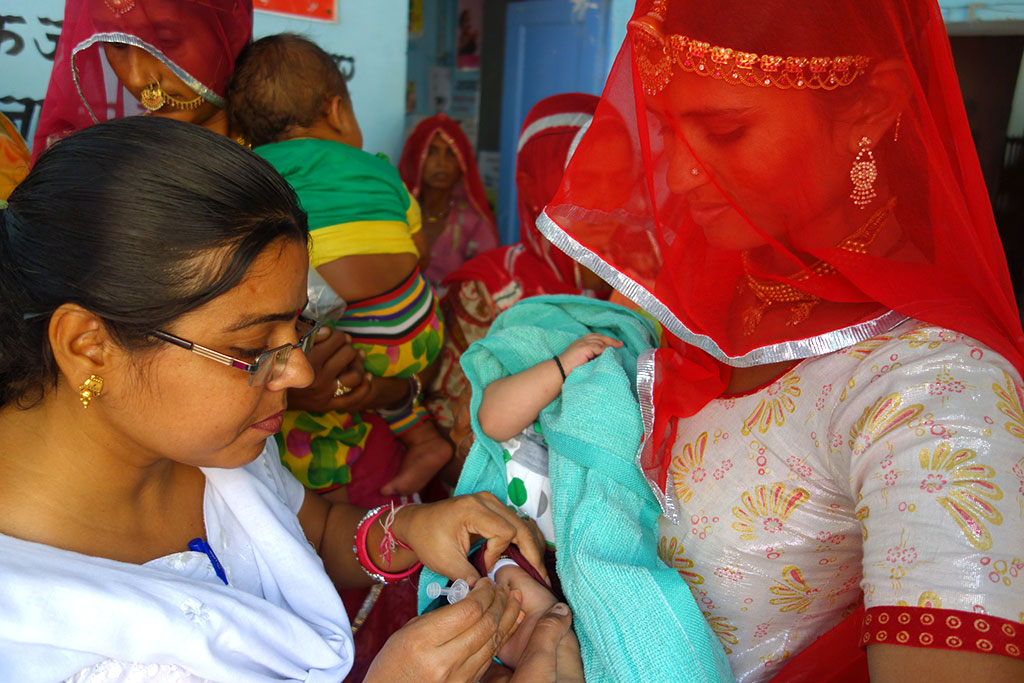
This blog originally appeared on the Vaccineswork website.
The introduction of a new vaccine to a country is not a one-time event—it’s a process.
At USAID’s flagship Maternal and Child Survival Program (MCSP), we know first-hand the extensive technical support required to introduce new and underutilized vaccines. It requires time and energy, resources, and continuous monitoring—and it’s a process that can’t be undertaken alone. To be successful, introducing a new vaccine involves partnerships with local and national government, as well as the communities being served.
In support of USAID’s efforts to help vaccinate 300 million children by 2020, MCSP provides focused technical assistance to prepare countries for the smooth and successful introduction of new, lifesaving vaccines. When a country eligible for Gavi support (typically those with low income) decides to introduce a new vaccine, several tasks must be completed:
- Secure partnerships—including government buy-in—and adequate resources;
- Achieve consensus on all planned activities;
- Ensure coordination around the process between the Ministry of Health, Ministry of Finance, and Interagency Coordinating Committee (ICC);
- Upgrade cold chain storage capacity to accommodate both traditional and additional new vaccines;
- Train health workers and community volunteers to deliver an increased number of vaccines at the health facility level, and to manage and dispose medical waste safely at immunization sites;
- Revise technical guidelines, recording and reporting management tools, and appropriate information, education & communication materials;
- Facilitate good communication with parents to address any vaccine-related concerns; and
- Establish a surveillance system prior to and after the new vaccine introduction.
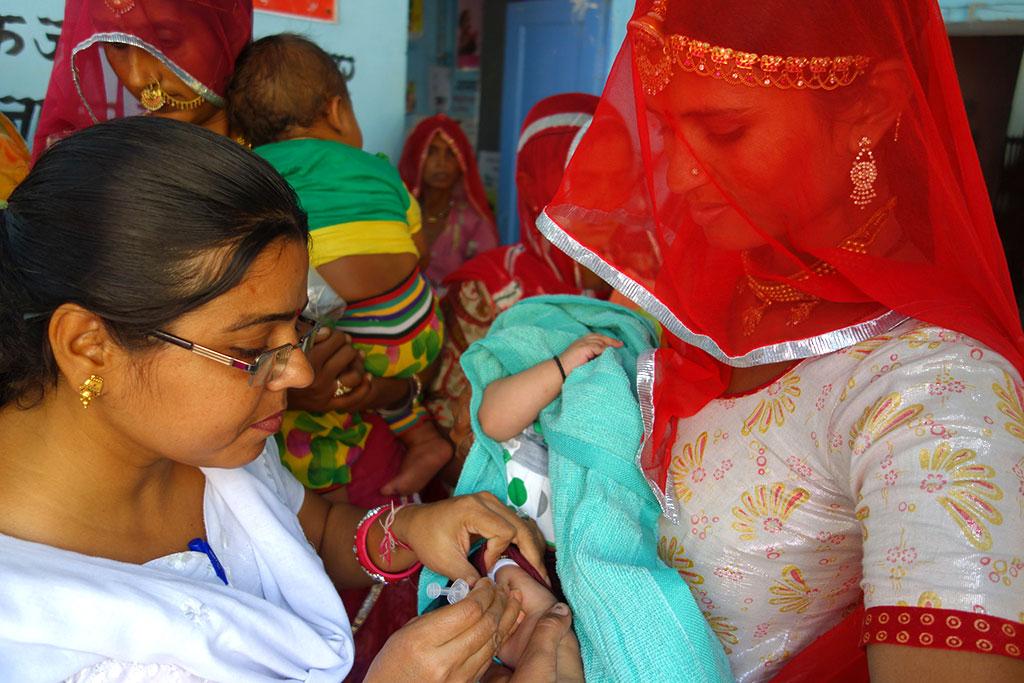
camp in Rajasthan, India. (Indrani Kashyap/Jhpiego)
Moreover, six steps are considered critical by all stakeholders when planning for new vaccine introduction:
- Step 1: Kick start decision making process at the country level
Ministry of Health Officials—with support from coordinating partners—decide on which vaccine to introduce and its presentation and formulation; - Step 2: Develop vaccine proposal
In-country World Health Organization Expanded Program on Immunization technical staff and coordinating partners plan key documents, complete the application form to Gavi, and revise policy documents to include the new vaccine; - Step 3: Submit approved and signed proposal to Gavi
Government and ICC partners submit the proposal to the Gavi Secretariat. - Step 4: Prepare for new vaccine introduction
With support from partners, the Ministry of Health addresses technical, cold chain and logistics, communication, social mobilization and surveillance challenges. - Step 5: Enlist local authority to host vaccine launch ceremony
This event, which partners usually want to attend, must be well planned—it’s success is key to motivating the community to accept the vaccine. - Step 6: Vaccinate eligible children
Post introduction activities—such as monitoring and supervision—need to ensure all eligible children are immunized.

Mundri East County, South Sudan. (Kate Holt/Jhpiego)
Introducing a new vaccine to a country typically takes six to eight months to complete, and has been critical to the Program’s efforts to end preventable child deaths. A lot of attention is focused on the so-called “launch.” But we must remember that, after a long period of planning, the launch ceremony celebrates the start of a vaccine’s roll out—and not the end of the journey.
The culmination of this process shepherds in a new and critical phase in saving the lives of children: our work to strengthen immunization systems and programs to protect those most vulnerable against all vaccine preventable diseases. In more than 20 countries worldwide, MCSP aims every day to make access to—and widespread use of—services equitable as we dramatically reduce the burden of disease.